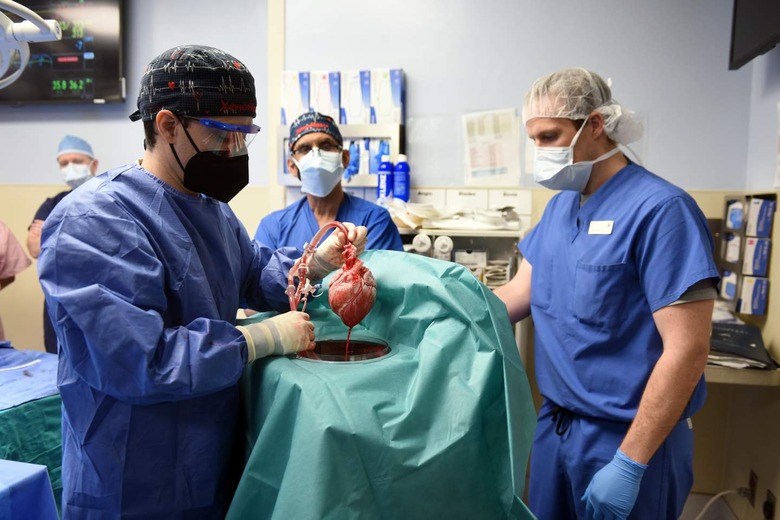
Terminally Ill U.S. Man Gets A Pig's Heart With A Successful Transplant
The organ donor crisis has long inspired the medical fraternity to look for feasible donors from other species. This week, doctors at the University of Maryland School of Medicine (UMSOM) have successfully executed the world's first heterologous heart transplant, placing a genetically-modified pig's heart into a terminally ill human being. Note: This article contains imagery and descriptions of human and animal surgery and xenotransplantation. Some readers may find this content upsetting.
Image: Coryn/Shutterstock
In a press note, the University of Maryland Medical Center (UMMC) announced this was the only option available to David Bennett, the 57-year patient who had been dependent on a lung and heart bypass machine — called extracorporeal membrane oxygenation (ECMO) — for his survival. He was admitted to the hospital six weeks prior because of a "life-threatening arrhythmia (irregular heart beat)."
Bennett neither qualified for a transplant using a heart from a human heart nor a mechanical heart due to his heart's condition and medical history and that left xenotransplantation as his only option. Xenotransplantation, as the World Health Organization defines, is a transplant of living cells, tissues, and organs from one species to another. As expected, it is typically used to define the transplant of animal organs to humans.
Pig's heart genetically modified to improve acceptance inside human body
The US Food and Drug Administration (FDA) approved the emergency transplant using a pig's heart by using its provision for expanded access (compassionate use). This provision is only valid when a terminally ill patient has no other option but to go for an experimental treatment procedure like this one.
Xenotransplantation poses the problem of triggering an autoimmune reaction in response to a foreign organ being introduced in the human body. To mitigate the effects, six human genes were selectively introduced into the genome to improve the chances of the patient's immune system accepting the pig's heart. Meanwhile, three genes that could cause quick rejection of pig organs by antibodies in humans were "knocked out" in the pig whereas one more gene was knocked out to prevent the pig's heart from growing excessively while in Bennett's body. Bennett also got an experimental drug alongside the genetic modifications to improve chances of his body accepting the heart.
In its announcement, UMSOM confirmed the patient has been stable for three days following the surgery. His condition would be monitored over the coming weeks to evaluate the effects of the non-human heart.
Pigs, the most credible non-human candidate for organ transplants
Pig organs have been considered for transplants in humans because of their similarity with human organs. The pig heart for this surgery was provided by a company named Revivicor which works on regenerative medical procedures. The pig-human heart transplant was part of the $15.7 million grant from Revivicor to UMSOM to study the effects of xenotransplantation using genetically-modified pig hearts, called "UHearts," by the company. Traditionally these UHearts have been used for experimental transplants in baboon, with this being the first one ever tried on a human.
Although xenotransplantation has been attempted for over a century but have sparked ethical debates and generated interest than expected — despite the dearth of human donors as pointed by the US government's organ donor portal organdonor.gov — for the risk of organs being rejected due to immunological differences. Humans share 99% of the DNA with chimpanzees, which is why the latter were previously considered for organ transplants. But the research has now shifted towards pigs because chimpanzees are now classified as endangered species. In September 2021, a kidney transplant was performed successfully using a kidney from a "genetically engineered pig" at NYU Langone Health.
Conversely, human organs are often used in other animals to study the working of organs — although with some resistance on ethical grounds. For instance, kidneys from aborted human fetuses have been transplanted in mice (via CBS News) for them to be grown bigger and used for a human transplant later. Human tumors have also been inserted in mice to test the manifestation of the tumor.