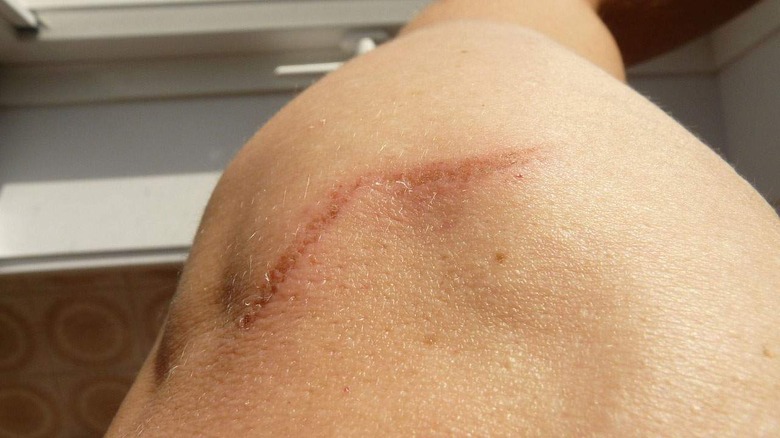
Researchers Discover A Way To Make Wounds Heal Without Forming Scar Tissue
A group of researchers has been conducting work in an attempt to figure out why we form scar tissue after healing from surgery or an injury. Many people don't like the appearance of scars, and scar tissue is weaker and lacks some of the functionality of normal skin. For instance, scar tissue has no hair follicles, no sweat glands, and is inflexible.
Scars can also limit the body's ability to move and adapt to changing temperatures. After years of research, scientists have discovered that interfering with some molecular signals during healing can produce tissue indistinguishable from normal skin. According to researcher Michael Longaker, scars form because they can seal an opening in the skin more quickly than normal skin can grow.
The problem with sealing the skin slowly is that the person can get an infection or potentially bleed to death. Longaker and colleagues on the research program discovered that tension during skin repair played a critical role in scarring. The team focused on a gene called engrailed that helps create a protein sometimes found in fibroblasts, a type of skin cell that drives scarring.
In experiments conducted in mice, the team discovered that a subpopulation of fibroblast cells in the skin that don't normally express engrailed began to do so during scarring. The team also looked at the role mechanical stress might play in turning on the engrailed gene. Researchers looked at mouse fibroblast cells that did not express engrailed and grew them in the laboratory in three different environments.
The environments included a soft gel that didn't produce mechanical strain on the growing fibroblasts, a stiff plastic dish that produces mechanical strain, and the same plastic disc but with a chemical blocking mechanical-strain signaling. Fibroblasts grown on the tension-free gel did not express engrailed, but those grown on the plastic disc did. The cells in the disc treated with the chemical did not express engrailed.
The team identified a drug called verteporfin that is approved by the FDA already as a treatment for eye disease designed to block mechanical-stress signaling in cells. When they treated mice wounds with the drug, normal skin sealed the wounds. The next stage in the research will be preclinical work on other animals, and if the results are successful, a clinical trial could follow.