Researchers Create Strangely Shaped Liquids Using Electric Fields
We've all seen drops of water and know that they tend to take on the same basic shape no matter what liquid the drop is made from. Researchers from Aalto University in Finland wanted to disrupt what's known as thermodynamic equilibrium to explore what happens and to determine if they could control what happened when equilibrium was disrupted.
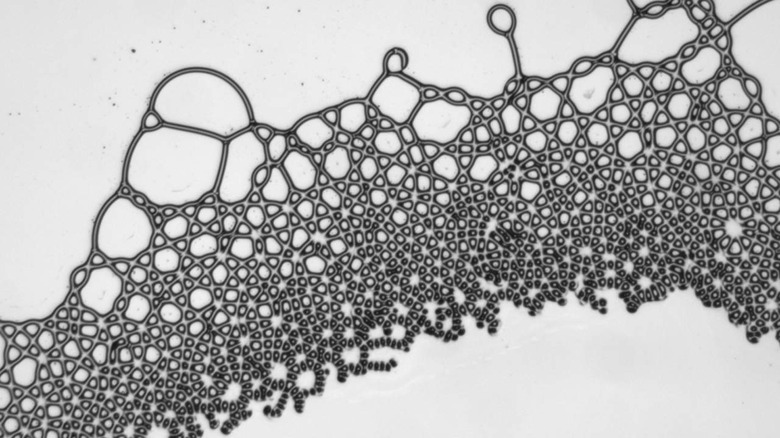
For the study, researchers worked with a combination of oils featuring different dielectric constants and levels of conductivity. Although the liquids were subjected to an electric field and the researchers discovered they could create droplets shaped like squares and liquid lattices.
Dr. Nikos Kyriakopoulos said when the team turned on the electric field over their mixture an electric charge would accumulate at the interface between the oils. Charge density was able to push the material out of thermodynamic equilibrium, creating unique formations. For the experiment, the liquids were confined in thin and nearly two-dimensional sheets. Researchers discovered their process forces the liquids to reshape into unexpected droplets and patterns.
The technique created droplets made into squares and hexagons with straight sides, both nearly impossible to create naturally. In nature, droplets and bubbles typically form spheres. Researchers were also successful in forcing two different liquids to create an interconnected lattice.
Lattices are common in the natural world for solid materials but are not seen in liquids. One of the most complicated shapes project researchers created was a torus, which is a donut shape. That uncommon shape was stable as long as the electric field was applied to the liquid. In nature, liquids naturally collapse in to fill holes at their center. Critically discovered in the experiment was that the shapes were sustainable due to the motion of the electrical charge at the interface, preventing them from collapsing into equilibrium.