FDA Authorizes A Unique Daytime Gadget To Treat Snoring And Sleep Apnea
The FDA has announced marketing authorization for the eXciteOSA device, a unique gadget that is used during the day to help reduce snoring and sleep apnea at night. Ordinarily, both nighttime issues are treated using devices that the patient wears while asleep. eXciteOSA is different: it's a device you use during the day to stimulate the tongue muscle.
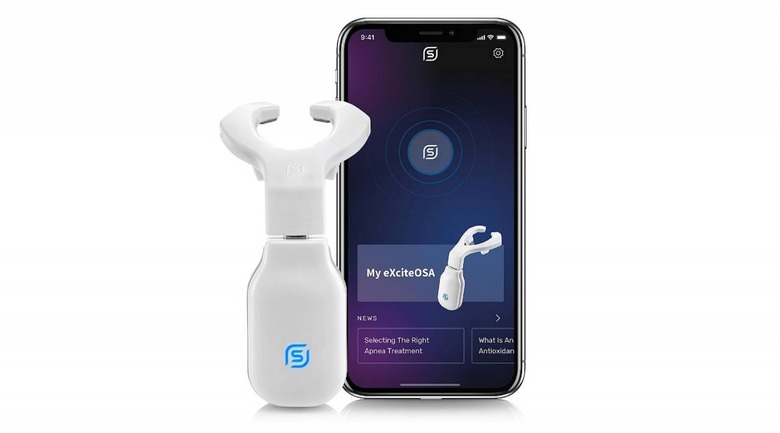
This device is authorized as a prescription treatment for mild obstructive sleep apnea, a condition in which a person may briefly stop breathing or breathe shallowly multiple times a night, as well as snoring. eXciteOSA is, as the picture below shows, a device that is placed over the tongue during the day.

The device delivers neuromuscular stimulation using four electrodes, two above and two below the tongue. Patients use the device for 20 minutes daily for the first six weeks, then only once a week after that. The treatment is meant for adults ages 18 and older who don't have contraindicated issues and dental features.
As well, the device isn't meant for individuals who have severe obstructive sleep apnea. In a statement about the authorization, FDA Director of the Office of Ophthalmic, Anesthesia, Respiratory, ENT and Dental Devices Dr. Malvina Eydelman said:
Obstructive sleep apnea not only impacts sleep quality but can have other serious health impacts if untreated. Today's authorization offers a new option for the thousands of individuals who experience snoring or mild sleep apnea.