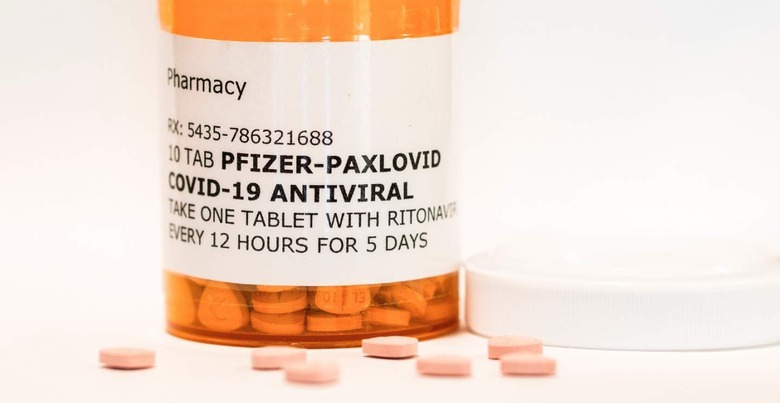COVID-19 Antiviral Pill Gets FDA Authorization: Here's The Small Print
Pfizer's Paxlovid is the first oral COVID-19 treatment to get FDA authorization for use in the US. The health agency announced its decision today, noting that it is only available for certain patients. Though Paxlovid won't protect you from developing COVID-19, taking it early enough may prevent the infection from becoming severe.
What's Paxlovid?
Paxlovid is a pill that patients take by mouth, adding a new option to the growing — but still slim — arsenal of medicine developed to address COVID-19. Pfizer published details on its drug in early November, revealing a notable decrease in hospitalization and death risk among patients who were given Paxlovid soon after testing positive.
The new oral antiviral medication is what's known as a SARS-CoV-2-3CL protease inhibitor, according to the pharmaceutical company. The drug works by blocking an enzyme the virus needs in order to replicate; the inhibitor is dosed alongside ritonavir, an antiretroviral medication, which works to slow the breakdown of the proteases inhibitor.
By combining the two drugs, Pfizer notes, the inhibitor can remain active in the patient's body for a longer period of time — and at greater concentrations — in order to slow down how fast the virus replicates. This, in turn, can help ensure one's illness doesn't progress to a more severe stage, though its efficacy is based on taking the drug within the first few days of testing positive.
Eligible patients
The FDA's Paxlovid authorization only covers certain patients, the agency explained today. The antiviral drug can be given to kids ages 12 and older who weigh at least 88lbs, as well as adults, if they've tested positive for COVID-19 and still only have mild to moderate symptoms. These patients, the FDA notes, must be considered high-risk for severe disease outcomes to get the drug.
The drug is meant to be administered within the first five days of symptoms and is taken twice a day over the course of five days. Those infected with COVID-19 can only get the drug as a prescription, with the biggest appeal being its simplicity: someone may be able to take this drug at home soon after testing positive, potentially avoiding hospitalization altogether.
The FDA stresses that Paxlovid isn't a substitute for preventative measures, namely getting the primary series of COVID-19 vaccines and, when eligible, the booster shot. The EUA was granted amid the rapid spread of omicron, the latest SARS-CoV-2 variant of concern first identified in South Africa.